 Found 180 hits from Novartis Institute of Biomedical Research
Found 180 hits from Novartis Institute of Biomedical Research Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Proto-oncogene tyrosine-protein kinase Src
(Homo sapiens (Human)) | BDBM50320214
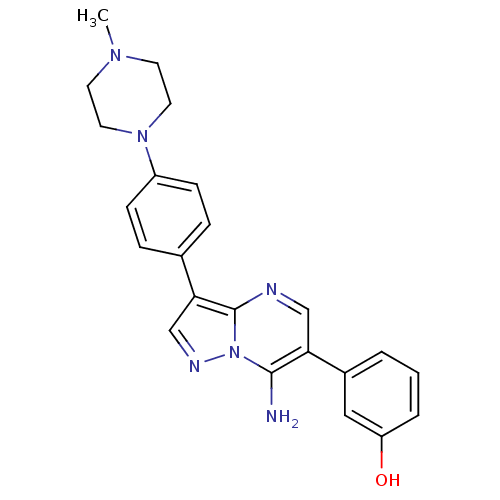
(3-(7-amino-3-(4-(4-methylpiperazin-1-yl)phenyl)pyr...)Show SMILES CN1CCN(CC1)c1ccc(cc1)-c1cnn2c(N)c(cnc12)-c1cccc(O)c1 Show InChI InChI=1S/C23H24N6O/c1-27-9-11-28(12-10-27)18-7-5-16(6-8-18)21-15-26-29-22(24)20(14-25-23(21)29)17-3-2-4-19(30)13-17/h2-8,13-15,30H,9-12,24H2,1H3 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of SRC |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320205
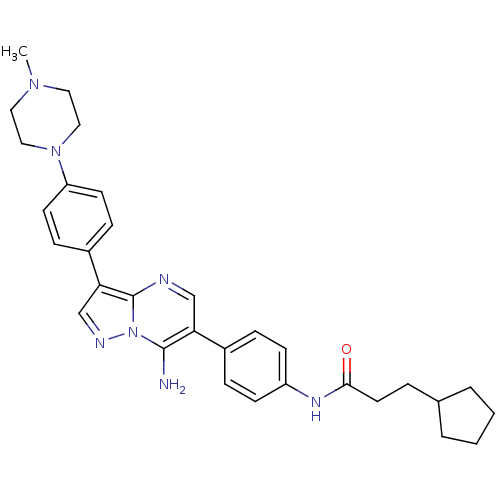
(CHEMBL1083001 | N-(4-(7-amino-3-(4-(4-methylpipera...)Show SMILES CN1CCN(CC1)c1ccc(cc1)-c1cnn2c(N)c(cnc12)-c1ccc(NC(=O)CCC2CCCC2)cc1 Show InChI InChI=1S/C31H37N7O/c1-36-16-18-37(19-17-36)26-13-9-24(10-14-26)28-21-34-38-30(32)27(20-33-31(28)38)23-7-11-25(12-8-23)35-29(39)15-6-22-4-2-3-5-22/h7-14,20-22H,2-6,15-19,32H2,1H3,(H,35,39) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320225
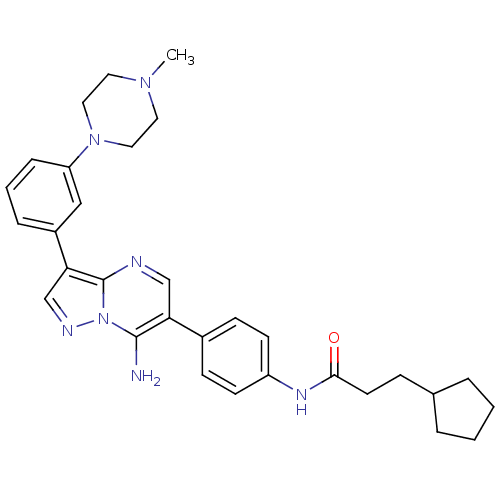
(CHEMBL1085316 | N-(4-(7-amino-3-(3-(4-methylpipera...)Show SMILES CN1CCN(CC1)c1cccc(c1)-c1cnn2c(N)c(cnc12)-c1ccc(NC(=O)CCC2CCCC2)cc1 Show InChI InChI=1S/C31H37N7O/c1-36-15-17-37(18-16-36)26-8-4-7-24(19-26)28-21-34-38-30(32)27(20-33-31(28)38)23-10-12-25(13-11-23)35-29(39)14-9-22-5-2-3-6-22/h4,7-8,10-13,19-22H,2-3,5-6,9,14-18,32H2,1H3,(H,35,39) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 4 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Homo sapiens (Human)) | BDBM50135541
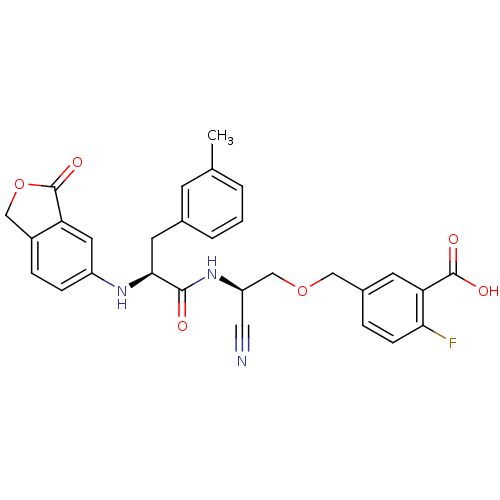
(5-{(R)-2-Cyano-2-[(S)-2-(3-oxo-1,3-dihydro-isobenz...)Show SMILES Cc1cccc(C[C@H](Nc2ccc3COC(=O)c3c2)C(=O)N[C@@H](COCc2ccc(F)c(c2)C(O)=O)C#N)c1 Show InChI InChI=1S/C29H26FN3O6/c1-17-3-2-4-18(9-17)11-26(32-21-7-6-20-15-39-29(37)23(20)12-21)27(34)33-22(13-31)16-38-14-19-5-8-25(30)24(10-19)28(35)36/h2-10,12,22,26,32H,11,14-16H2,1H3,(H,33,34)(H,35,36)/t22-,26+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 4.10 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibtitory activity against cathepsin B (catB) |
Bioorg Med Chem Lett 13: 4121-4 (2003)
BindingDB Entry DOI: 10.7270/Q2BC3XZC |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Homo sapiens (Human)) | BDBM50135543

(3-{(R)-2-Cyano-2-[(S)-2-(2-methyl-1,3-dioxo-2,3-di...)Show SMILES CN1C(=O)c2ccc(N[C@@H](Cc3cccc(C)c3)C(=O)N[C@@H](COCc3cccc(c3)C(O)=O)C#N)cc2C1=O Show InChI InChI=1S/C30H28N4O6/c1-18-5-3-6-19(11-18)13-26(32-22-9-10-24-25(14-22)29(37)34(2)28(24)36)27(35)33-23(15-31)17-40-16-20-7-4-8-21(12-20)30(38)39/h3-12,14,23,26,32H,13,16-17H2,1-2H3,(H,33,35)(H,38,39)/t23-,26+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 4.90 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibtitory activity against cathepsin B (catB) |
Bioorg Med Chem Lett 13: 4121-4 (2003)
BindingDB Entry DOI: 10.7270/Q2BC3XZC |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Homo sapiens (Human)) | BDBM50135533

((S)-N-[(R)-Cyano-(3-tetrazol-1-yl-benzyloxymethyl)...)Show SMILES Cc1cccc(C[C@H](Nc2ccc3COC(=O)c3c2)C(=O)N[C@@H](COCc2cccc(c2)-n2cnnn2)C#N)c1 Show InChI InChI=1S/C29H27N7O4/c1-19-4-2-5-20(10-19)12-27(32-23-9-8-22-16-40-29(38)26(22)13-23)28(37)33-24(14-30)17-39-15-21-6-3-7-25(11-21)36-18-31-34-35-36/h2-11,13,18,24,27,32H,12,15-17H2,1H3,(H,33,37)/t24-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibtitory activity against cathepsin B (catB) |
Bioorg Med Chem Lett 13: 4121-4 (2003)
BindingDB Entry DOI: 10.7270/Q2BC3XZC |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320209

(1-(4-(7-amino-3-(4-(4-methylpiperazin-1-yl)phenyl)...)Show SMILES CN1CCN(CC1)c1ccc(cc1)-c1cnn2c(N)c(cnc12)-c1ccc(NC(=O)NCC2CCCCC2)cc1 Show InChI InChI=1S/C31H38N8O/c1-37-15-17-38(18-16-37)26-13-9-24(10-14-26)28-21-35-39-29(32)27(20-33-30(28)39)23-7-11-25(12-8-23)36-31(40)34-19-22-5-3-2-4-6-22/h7-14,20-22H,2-6,15-19,32H2,1H3,(H2,34,36,40) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Homo sapiens (Human)) | BDBM50135535
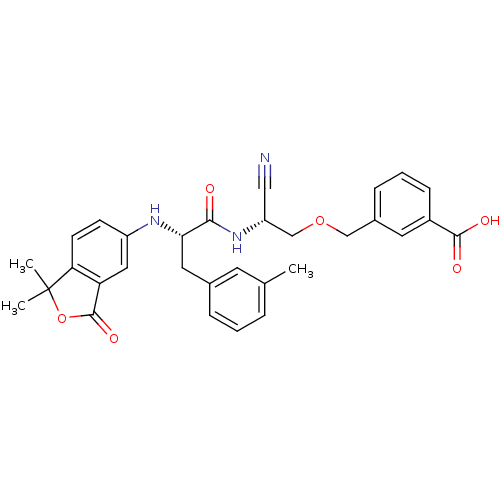
(3-{(R)-2-Cyano-2-[(S)-2-(1,1-dimethyl-3-oxo-1,3-di...)Show SMILES Cc1cccc(C[C@H](Nc2ccc3c(c2)C(=O)OC3(C)C)C(=O)N[C@@H](COCc2cccc(c2)C(O)=O)C#N)c1 Show InChI InChI=1S/C31H31N3O6/c1-19-6-4-7-20(12-19)14-27(33-23-10-11-26-25(15-23)30(38)40-31(26,2)3)28(35)34-24(16-32)18-39-17-21-8-5-9-22(13-21)29(36)37/h4-13,15,24,27,33H,14,17-18H2,1-3H3,(H,34,35)(H,36,37)/t24-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 5.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibtitory activity against cathepsin B (catB) |
Bioorg Med Chem Lett 13: 4121-4 (2003)
BindingDB Entry DOI: 10.7270/Q2BC3XZC |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Homo sapiens (Human)) | BDBM50135537

(3-{(R)-2-Cyano-2-[(S)-2-(3-oxo-indan-5-ylamino)-3-...)Show SMILES Cc1cccc(C[C@H](Nc2ccc3CCC(=O)c3c2)C(=O)N[C@@H](COCc2cccc(c2)C(O)=O)C#N)c1 Show InChI InChI=1S/C30H29N3O5/c1-19-4-2-5-20(12-19)14-27(32-24-10-8-22-9-11-28(34)26(22)15-24)29(35)33-25(16-31)18-38-17-21-6-3-7-23(13-21)30(36)37/h2-8,10,12-13,15,25,27,32H,9,11,14,17-18H2,1H3,(H,33,35)(H,36,37)/t25-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 5.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibtitory activity against cathepsin B (catB) |
Bioorg Med Chem Lett 13: 4121-4 (2003)
BindingDB Entry DOI: 10.7270/Q2BC3XZC |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320224
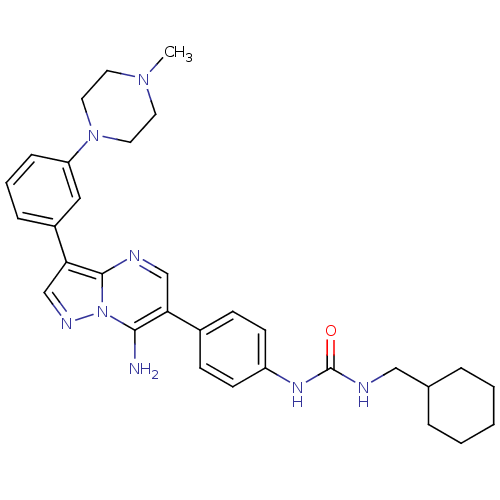
(1-(4-(7-amino-3-(3-(4-methylpiperazin-1-yl)phenyl)...)Show SMILES CN1CCN(CC1)c1cccc(c1)-c1cnn2c(N)c(cnc12)-c1ccc(NC(=O)NCC2CCCCC2)cc1 Show InChI InChI=1S/C31H38N8O/c1-37-14-16-38(17-15-37)26-9-5-8-24(18-26)28-21-35-39-29(32)27(20-33-30(28)39)23-10-12-25(13-11-23)36-31(40)34-19-22-6-3-2-4-7-22/h5,8-13,18,20-22H,2-4,6-7,14-17,19,32H2,1H3,(H2,34,36,40) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 6 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Homo sapiens (Human)) | BDBM50135546
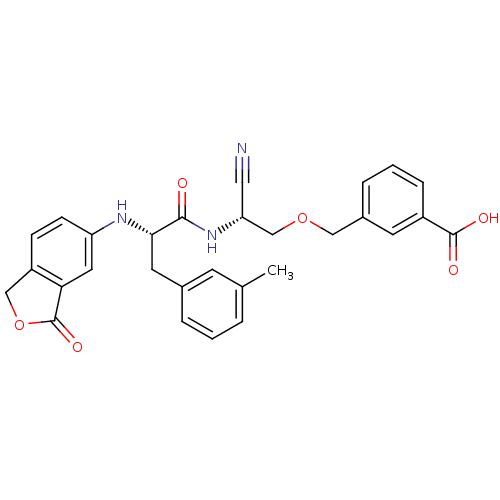
(3-{(R)-2-Cyano-2-[(S)-2-(3-oxo-1,3-dihydro-isobenz...)Show SMILES Cc1cccc(C[C@H](Nc2ccc3COC(=O)c3c2)C(=O)N[C@@H](COCc2cccc(c2)C(O)=O)C#N)c1 Show InChI InChI=1S/C29H27N3O6/c1-18-4-2-5-19(10-18)12-26(31-23-9-8-22-16-38-29(36)25(22)13-23)27(33)32-24(14-30)17-37-15-20-6-3-7-21(11-20)28(34)35/h2-11,13,24,26,31H,12,15-17H2,1H3,(H,32,33)(H,34,35)/t24-,26+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 6.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibtitory activity against cathepsin B (catB) |
Bioorg Med Chem Lett 13: 4121-4 (2003)
BindingDB Entry DOI: 10.7270/Q2BC3XZC |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320216
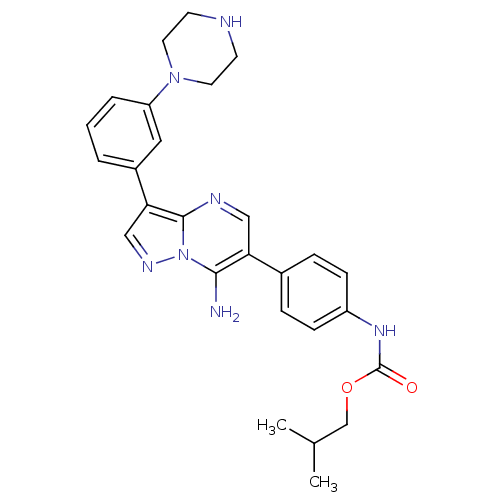
(CHEMBL1083644 | isobutyl4-(7-amino-3-(3-(piperazin...)Show SMILES CC(C)COC(=O)Nc1ccc(cc1)-c1cnc2c(cnn2c1N)-c1cccc(c1)N1CCNCC1 Show InChI InChI=1S/C27H31N7O2/c1-18(2)17-36-27(35)32-21-8-6-19(7-9-21)23-15-30-26-24(16-31-34(26)25(23)28)20-4-3-5-22(14-20)33-12-10-29-11-13-33/h3-9,14-16,18,29H,10-13,17,28H2,1-2H3,(H,32,35) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK in human Jurkat cells assessed as ZAP70 phosphorylation by FACS assay |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320218
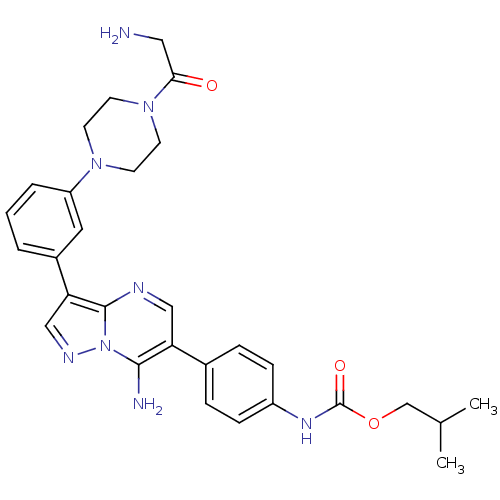
(CHEMBL1082404 | isobutyl4-(7-amino-3-(3-(4-(2-amin...)Show SMILES CC(C)COC(=O)Nc1ccc(cc1)-c1cnc2c(cnn2c1N)-c1cccc(c1)N1CCN(CC1)C(=O)CN Show InChI InChI=1S/C29H34N8O3/c1-19(2)18-40-29(39)34-22-8-6-20(7-9-22)24-16-32-28-25(17-33-37(28)27(24)31)21-4-3-5-23(14-21)35-10-12-36(13-11-35)26(38)15-30/h3-9,14,16-17,19H,10-13,15,18,30-31H2,1-2H3,(H,34,39) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 7 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK in human Jurkat cells assessed as ZAP70 phosphorylation by FACS assay |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320223
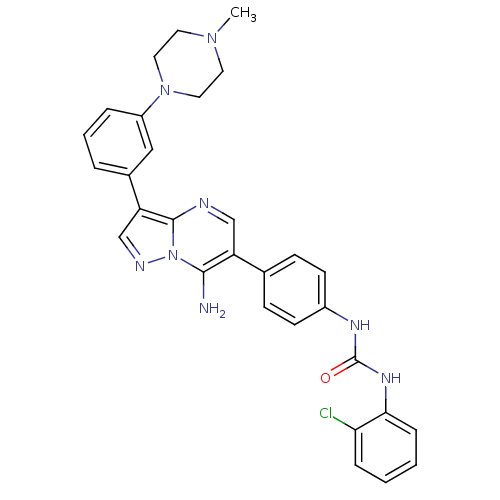
(1-(4-(7-amino-3-(3-(4-methylpiperazin-1-yl)phenyl)...)Show SMILES CN1CCN(CC1)c1cccc(c1)-c1cnn2c(N)c(cnc12)-c1ccc(NC(=O)Nc2ccccc2Cl)cc1 Show InChI InChI=1S/C30H29ClN8O/c1-37-13-15-38(16-14-37)23-6-4-5-21(17-23)25-19-34-39-28(32)24(18-33-29(25)39)20-9-11-22(12-10-20)35-30(40)36-27-8-3-2-7-26(27)31/h2-12,17-19H,13-16,32H2,1H3,(H2,35,36,40) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320204

(3-(4-(7-amino-3-(3-(4-methylpiperazin-1-yl)phenyl)...)Show SMILES CN(C1CCCCC1)C(=O)Nc1ccc(cc1)-c1cnc2c(cnn2c1N)-c1cccc(c1)N1CCN(C)CC1 Show InChI InChI=1S/C31H38N8O/c1-36-15-17-38(18-16-36)26-10-6-7-23(19-26)28-21-34-39-29(32)27(20-33-30(28)39)22-11-13-24(14-12-22)35-31(40)37(2)25-8-4-3-5-9-25/h6-7,10-14,19-21,25H,3-5,8-9,15-18,32H2,1-2H3,(H,35,40) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase HCK
(Homo sapiens (Human)) | BDBM50320214

(3-(7-amino-3-(4-(4-methylpiperazin-1-yl)phenyl)pyr...)Show SMILES CN1CCN(CC1)c1ccc(cc1)-c1cnn2c(N)c(cnc12)-c1cccc(O)c1 Show InChI InChI=1S/C23H24N6O/c1-27-9-11-28(12-10-27)18-7-5-16(6-8-18)21-15-26-29-22(24)20(14-25-23(21)29)17-3-2-4-19(30)13-17/h2-8,13-15,30H,9-12,24H2,1H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of HCK |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320214

(3-(7-amino-3-(4-(4-methylpiperazin-1-yl)phenyl)pyr...)Show SMILES CN1CCN(CC1)c1ccc(cc1)-c1cnn2c(N)c(cnc12)-c1cccc(O)c1 Show InChI InChI=1S/C23H24N6O/c1-27-9-11-28(12-10-27)18-7-5-16(6-8-18)21-15-26-29-22(24)20(14-25-23(21)29)17-3-2-4-19(30)13-17/h2-8,13-15,30H,9-12,24H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 8 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK in human Jurkat cells assessed as ZAP70 phosphorylation by FACS assay |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Homo sapiens (Human)) | BDBM50135532
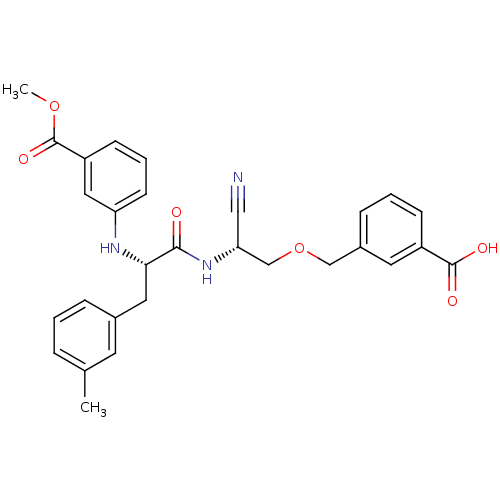
(3-{2-cyano-2-[1-(3-methyloxycarbonylanilino)-2-(3-...)Show SMILES COC(=O)c1cccc(N[C@@H](Cc2cccc(C)c2)C(=O)N[C@@H](COCc2cccc(c2)C(O)=O)C#N)c1 Show InChI InChI=1S/C29H29N3O6/c1-19-6-3-7-20(12-19)14-26(31-24-11-5-10-23(15-24)29(36)37-2)27(33)32-25(16-30)18-38-17-21-8-4-9-22(13-21)28(34)35/h3-13,15,25-26,31H,14,17-18H2,1-2H3,(H,32,33)(H,34,35)/t25-,26+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 8.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibtitory activity against cathepsin B (catB) |
Bioorg Med Chem Lett 13: 4121-4 (2003)
BindingDB Entry DOI: 10.7270/Q2BC3XZC |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Homo sapiens (Human)) | BDBM50135544

(2-Chloro-5-[(R)-2-cyano-2-((S)-2-phenylamino-3-m-t...)Show SMILES Cc1cccc(C[C@H](Nc2ccccc2)C(=O)N[C@@H](COCc2ccc(Cl)c(c2)C(O)=O)C#N)c1 Show InChI InChI=1S/C27H26ClN3O4/c1-18-6-5-7-19(12-18)14-25(30-21-8-3-2-4-9-21)26(32)31-22(15-29)17-35-16-20-10-11-24(28)23(13-20)27(33)34/h2-13,22,25,30H,14,16-17H2,1H3,(H,31,32)(H,33,34)/t22-,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 8.70 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibtitory activity against cathepsin B (catB) |
Bioorg Med Chem Lett 13: 4121-4 (2003)
BindingDB Entry DOI: 10.7270/Q2BC3XZC |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Homo sapiens (Human)) | BDBM50135547

(3-{(R)-2-Cyano-2-[(S)-2-(2-methyl-3-oxo-2,3-dihydr...)Show SMILES CN1Cc2ccc(N[C@@H](Cc3cccc(C)c3)C(=O)N[C@@H](COCc3cccc(c3)C(O)=O)C#N)cc2C1=O Show InChI InChI=1S/C30H30N4O5/c1-19-5-3-6-20(11-19)13-27(32-24-10-9-23-16-34(2)29(36)26(23)14-24)28(35)33-25(15-31)18-39-17-21-7-4-8-22(12-21)30(37)38/h3-12,14,25,27,32H,13,16-18H2,1-2H3,(H,33,35)(H,37,38)/t25-,27+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 9.30 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibtitory activity against cathepsin B (catB) |
Bioorg Med Chem Lett 13: 4121-4 (2003)
BindingDB Entry DOI: 10.7270/Q2BC3XZC |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Homo sapiens (Human)) | BDBM50135534

(5-[(R)-2-Cyano-2-((S)-2-phenylamino-3-m-tolyl-prop...)Show SMILES Cc1cccc(C[C@H](Nc2ccccc2)C(=O)N[C@@H](COCc2ccc(F)c(c2)C(O)=O)C#N)c1 Show InChI InChI=1S/C27H26FN3O4/c1-18-6-5-7-19(12-18)14-25(30-21-8-3-2-4-9-21)26(32)31-22(15-29)17-35-16-20-10-11-24(28)23(13-20)27(33)34/h2-13,22,25,30H,14,16-17H2,1H3,(H,31,32)(H,33,34)/t22-,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 12 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibtitory activity against cathepsin B (catB) |
Bioorg Med Chem Lett 13: 4121-4 (2003)
BindingDB Entry DOI: 10.7270/Q2BC3XZC |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Homo sapiens (Human)) | BDBM50135545

(3-{(R)-2-Cyano-2-[(S)-2-(3-methanesulfonyl-phenyla...)Show SMILES Cc1cccc(C[C@H](Nc2cccc(c2)S(C)(=O)=O)C(=O)N[C@@H](COCc2cccc(c2)C(O)=O)C#N)c1 Show InChI InChI=1S/C28H29N3O6S/c1-19-6-3-7-20(12-19)14-26(30-23-10-5-11-25(15-23)38(2,35)36)27(32)31-24(16-29)18-37-17-21-8-4-9-22(13-21)28(33)34/h3-13,15,24,26,30H,14,17-18H2,1-2H3,(H,31,32)(H,33,34)/t24-,26+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibtitory activity against cathepsin B (catB) |
Bioorg Med Chem Lett 13: 4121-4 (2003)
BindingDB Entry DOI: 10.7270/Q2BC3XZC |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320210
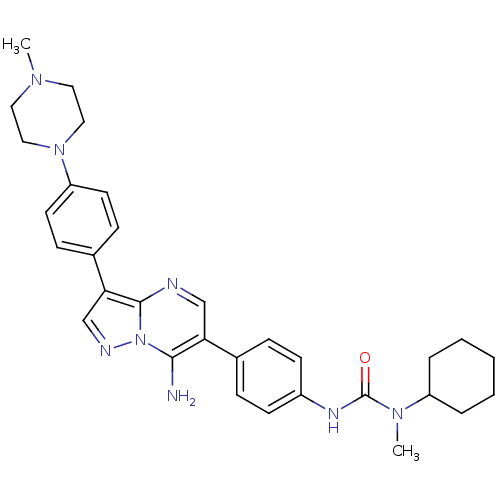
(3-(4-(7-amino-3-(4-(4-methylpiperazin-1-yl)phenyl)...)Show SMILES CN(C1CCCCC1)C(=O)Nc1ccc(cc1)-c1cnc2c(cnn2c1N)-c1ccc(cc1)N1CCN(C)CC1 Show InChI InChI=1S/C31H38N8O/c1-36-16-18-38(19-17-36)26-14-10-23(11-15-26)28-21-34-39-29(32)27(20-33-30(28)39)22-8-12-24(13-9-22)35-31(40)37(2)25-6-4-3-5-7-25/h8-15,20-21,25H,3-7,16-19,32H2,1-2H3,(H,35,40) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320209

(1-(4-(7-amino-3-(4-(4-methylpiperazin-1-yl)phenyl)...)Show SMILES CN1CCN(CC1)c1ccc(cc1)-c1cnn2c(N)c(cnc12)-c1ccc(NC(=O)NCC2CCCCC2)cc1 Show InChI InChI=1S/C31H38N8O/c1-37-15-17-38(18-16-37)26-13-9-24(10-14-26)28-21-35-39-29(32)27(20-33-30(28)39)23-7-11-25(12-8-23)36-31(40)34-19-22-5-3-2-4-6-22/h7-14,20-22H,2-6,15-19,32H2,1H3,(H2,34,36,40) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 13 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK in human Jurkat cells assessed as ZAP70 phosphorylation by FACS assay |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase HCK
(Homo sapiens (Human)) | BDBM50320205

(CHEMBL1083001 | N-(4-(7-amino-3-(4-(4-methylpipera...)Show SMILES CN1CCN(CC1)c1ccc(cc1)-c1cnn2c(N)c(cnc12)-c1ccc(NC(=O)CCC2CCCC2)cc1 Show InChI InChI=1S/C31H37N7O/c1-36-16-18-37(19-17-36)26-13-9-24(10-14-26)28-21-34-38-30(32)27(20-33-31(28)38)23-7-11-25(12-8-23)35-29(39)15-6-22-4-2-3-5-22/h7-14,20-22H,2-6,15-19,32H2,1H3,(H,35,39) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of HCK |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320207
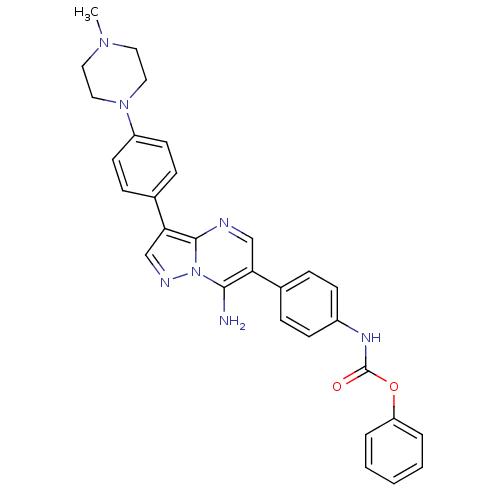
(CHEMBL1085512 | phenyl4-(7-amino-3-(4-(4-methylpip...)Show SMILES CN1CCN(CC1)c1ccc(cc1)-c1cnn2c(N)c(cnc12)-c1ccc(NC(=O)Oc2ccccc2)cc1 Show InChI InChI=1S/C30H29N7O2/c1-35-15-17-36(18-16-35)24-13-9-22(10-14-24)27-20-33-37-28(31)26(19-32-29(27)37)21-7-11-23(12-8-21)34-30(38)39-25-5-3-2-4-6-25/h2-14,19-20H,15-18,31H2,1H3,(H,34,38) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 14 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320222
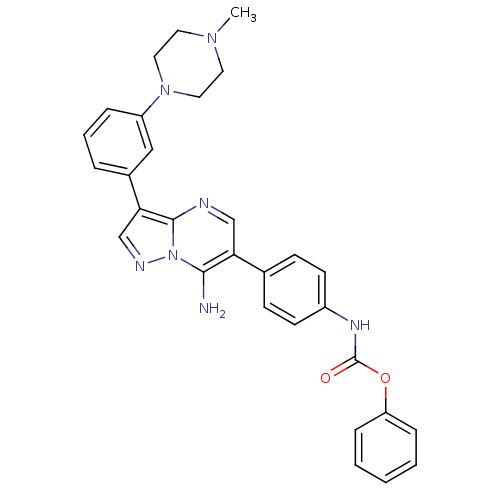
(CHEMBL1085564 | phenyl4-(7-amino-3-(3-(4-methylpip...)Show SMILES CN1CCN(CC1)c1cccc(c1)-c1cnn2c(N)c(cnc12)-c1ccc(NC(=O)Oc2ccccc2)cc1 Show InChI InChI=1S/C30H29N7O2/c1-35-14-16-36(17-15-35)24-7-5-6-22(18-24)27-20-33-37-28(31)26(19-32-29(27)37)21-10-12-23(13-11-21)34-30(38)39-25-8-3-2-4-9-25/h2-13,18-20H,14-17,31H2,1H3,(H,34,38) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320224

(1-(4-(7-amino-3-(3-(4-methylpiperazin-1-yl)phenyl)...)Show SMILES CN1CCN(CC1)c1cccc(c1)-c1cnn2c(N)c(cnc12)-c1ccc(NC(=O)NCC2CCCCC2)cc1 Show InChI InChI=1S/C31H38N8O/c1-37-14-16-38(17-15-37)26-9-5-8-24(18-26)28-21-35-39-29(32)27(20-33-30(28)39)23-10-12-25(13-11-23)36-31(40)34-19-22-6-3-2-4-7-22/h5,8-13,18,20-22H,2-4,6-7,14-17,19,32H2,1H3,(H2,34,36,40) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK in human Jurkat cells assessed as ZAP70 phosphorylation by FACS assay |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Homo sapiens (Human)) | BDBM50135548

((S)-N-[(R)-Cyano-(3-tetrazol-1-yl-benzyloxymethyl)...)Show SMILES Cc1cccc(C[C@H](Nc2ccccc2)C(=O)N[C@@H](COCc2cccc(c2)-n2cnnn2)C#N)c1 Show InChI InChI=1S/C27H27N7O2/c1-20-7-5-8-21(13-20)15-26(30-23-10-3-2-4-11-23)27(35)31-24(16-28)18-36-17-22-9-6-12-25(14-22)34-19-29-32-33-34/h2-14,19,24,26,30H,15,17-18H2,1H3,(H,31,35)/t24-,26+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| PubMed
| n/a | n/a | 17 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibtitory activity against cathepsin B (catB) |
Bioorg Med Chem Lett 13: 4121-4 (2003)
BindingDB Entry DOI: 10.7270/Q2BC3XZC |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase HCK
(Homo sapiens (Human)) | BDBM50320209

(1-(4-(7-amino-3-(4-(4-methylpiperazin-1-yl)phenyl)...)Show SMILES CN1CCN(CC1)c1ccc(cc1)-c1cnn2c(N)c(cnc12)-c1ccc(NC(=O)NCC2CCCCC2)cc1 Show InChI InChI=1S/C31H38N8O/c1-37-15-17-38(18-16-37)26-13-9-24(10-14-26)28-21-35-39-29(32)27(20-33-30(28)39)23-7-11-25(12-8-23)36-31(40)34-19-22-5-3-2-4-6-22/h7-14,20-22H,2-6,15-19,32H2,1H3,(H2,34,36,40) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 21 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of HCK |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320203

(CHEMBL1085511 | isobutyl4-(7-amino-3-(4-(4-methylp...)Show SMILES CC(C)COC(=O)Nc1ccc(cc1)-c1cnc2c(cnn2c1N)-c1ccc(cc1)N1CCN(C)CC1 Show InChI InChI=1S/C28H33N7O2/c1-19(2)18-37-28(36)32-22-8-4-20(5-9-22)24-16-30-27-25(17-31-35(27)26(24)29)21-6-10-23(11-7-21)34-14-12-33(3)13-15-34/h4-11,16-17,19H,12-15,18,29H2,1-3H3,(H,32,36) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 22 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320212
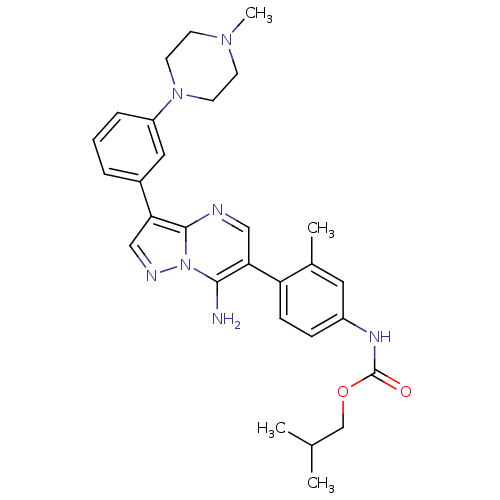
(CHEMBL1085996 | isobutyl4-(7-amino-3-(3-(4-methylp...)Show SMILES CC(C)COC(=O)Nc1ccc(c(C)c1)-c1cnc2c(cnn2c1N)-c1cccc(c1)N1CCN(C)CC1 Show InChI InChI=1S/C29H35N7O2/c1-19(2)18-38-29(37)33-22-8-9-24(20(3)14-22)26-16-31-28-25(17-32-36(28)27(26)30)21-6-5-7-23(15-21)35-12-10-34(4)11-13-35/h5-9,14-17,19H,10-13,18,30H2,1-4H3,(H,33,37) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK in human Jurkat cells assessed as ZAP70 phosphorylation by FACS assay |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320226

(CHEMBL1085240 | N-(4-(7-amino-3-(3-(4-methylpipera...)Show SMILES CN1CCN(CC1)c1cccc(c1)-c1cnn2c(N)c(cnc12)-c1ccc(NC(=O)c2ccc(cc2)C(F)(F)F)cc1 Show InChI InChI=1S/C31H28F3N7O/c1-39-13-15-40(16-14-39)25-4-2-3-22(17-25)27-19-37-41-28(35)26(18-36-29(27)41)20-7-11-24(12-8-20)38-30(42)21-5-9-23(10-6-21)31(32,33)34/h2-12,17-19H,13-16,35H2,1H3,(H,38,42) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 23 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320221
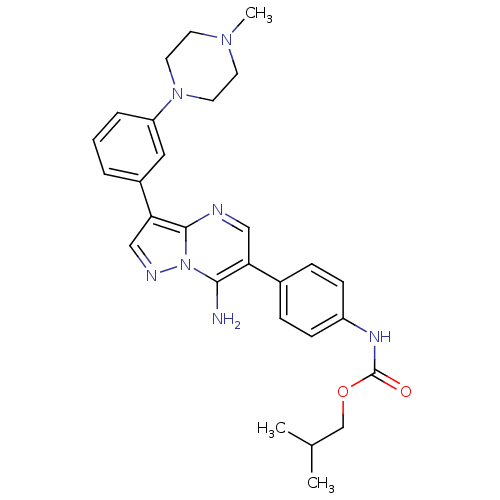
(CHEMBL1085317 | isobutyl4-(7-amino-3-(3-(4-methylp...)Show SMILES CC(C)COC(=O)Nc1ccc(cc1)-c1cnc2c(cnn2c1N)-c1cccc(c1)N1CCN(C)CC1 Show InChI InChI=1S/C28H33N7O2/c1-19(2)18-37-28(36)32-22-9-7-20(8-10-22)24-16-30-27-25(17-31-35(27)26(24)29)21-5-4-6-23(15-21)34-13-11-33(3)12-14-34/h4-10,15-17,19H,11-14,18,29H2,1-3H3,(H,32,36) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 24 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320208

(1-(4-(7-amino-3-(4-(4-methylpiperazin-1-yl)phenyl)...)Show SMILES CN1CCN(CC1)c1ccc(cc1)-c1cnn2c(N)c(cnc12)-c1ccc(NC(=O)Nc2ccccc2Cl)cc1 Show InChI InChI=1S/C30H29ClN8O/c1-37-14-16-38(17-15-37)23-12-8-21(9-13-23)25-19-34-39-28(32)24(18-33-29(25)39)20-6-10-22(11-7-20)35-30(40)36-27-5-3-2-4-26(27)31/h2-13,18-19H,14-17,32H2,1H3,(H2,35,36,40) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 25 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320229

(CHEMBL1084974 | N-(4-(7-amino-3-(3-(4-methylpipera...)Show SMILES CN1CCN(CC1)c1cccc(c1)-c1cnn2c(N)c(cnc12)-c1ccc(NS(=O)(=O)c2cccc(Cl)c2Cl)cc1 Show InChI InChI=1S/C29H27Cl2N7O2S/c1-36-12-14-37(15-13-36)22-5-2-4-20(16-22)24-18-34-38-28(32)23(17-33-29(24)38)19-8-10-21(11-9-19)35-41(39,40)26-7-3-6-25(30)27(26)31/h2-11,16-18,35H,12-15,32H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | 28 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK in human Jurkat cells assessed as ZAP70 phosphorylation by FACS assay |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320227
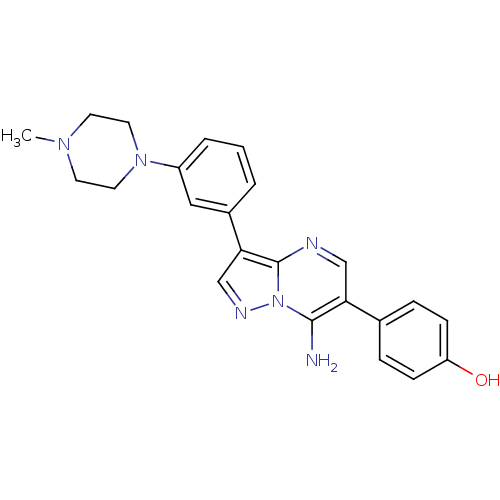
(4-(7-amino-3-(3-(4-methylpiperazin-1-yl)phenyl)pyr...)Show SMILES CN1CCN(CC1)c1cccc(c1)-c1cnn2c(N)c(cnc12)-c1ccc(O)cc1 Show InChI InChI=1S/C23H24N6O/c1-27-9-11-28(12-10-27)18-4-2-3-17(13-18)21-15-26-29-22(24)20(14-25-23(21)29)16-5-7-19(30)8-6-16/h2-8,13-15,30H,9-12,24H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK in human Jurkat cells assessed as ZAP70 phosphorylation by FACS assay |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320223

(1-(4-(7-amino-3-(3-(4-methylpiperazin-1-yl)phenyl)...)Show SMILES CN1CCN(CC1)c1cccc(c1)-c1cnn2c(N)c(cnc12)-c1ccc(NC(=O)Nc2ccccc2Cl)cc1 Show InChI InChI=1S/C30H29ClN8O/c1-37-13-15-38(16-14-37)23-6-4-5-21(17-23)25-19-34-39-28(32)24(18-33-29(25)39)20-9-11-22(12-10-20)35-30(40)36-27-8-3-2-7-26(27)31/h2-12,17-19H,13-16,32H2,1H3,(H2,35,36,40) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 31 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK in human Jurkat cells assessed as ZAP70 phosphorylation by FACS assay |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320225

(CHEMBL1085316 | N-(4-(7-amino-3-(3-(4-methylpipera...)Show SMILES CN1CCN(CC1)c1cccc(c1)-c1cnn2c(N)c(cnc12)-c1ccc(NC(=O)CCC2CCCC2)cc1 Show InChI InChI=1S/C31H37N7O/c1-36-15-17-37(18-16-36)26-8-4-7-24(19-26)28-21-34-38-30(32)27(20-33-31(28)38)23-10-12-25(13-11-23)35-29(39)14-9-22-5-2-3-6-22/h4,7-8,10-13,19-22H,2-3,5-6,9,14-18,32H2,1H3,(H,35,39) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 33 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK in human Jurkat cells assessed as ZAP70 phosphorylation by FACS assay |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320228

(4-(7-amino-3-(4-(4-methylpiperazin-1-yl)phenyl)pyr...)Show SMILES CN1CCN(CC1)c1ccc(cc1)-c1cnn2c(N)c(cnc12)-c1ccc(O)cc1 Show InChI InChI=1S/C23H24N6O/c1-27-10-12-28(13-11-27)18-6-2-17(3-7-18)21-15-26-29-22(24)20(14-25-23(21)29)16-4-8-19(30)9-5-16/h2-9,14-15,30H,10-13,24H2,1H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 36 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK in human Jurkat cells assessed as ZAP70 phosphorylation by FACS assay |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320207

(CHEMBL1085512 | phenyl4-(7-amino-3-(4-(4-methylpip...)Show SMILES CN1CCN(CC1)c1ccc(cc1)-c1cnn2c(N)c(cnc12)-c1ccc(NC(=O)Oc2ccccc2)cc1 Show InChI InChI=1S/C30H29N7O2/c1-35-15-17-36(18-16-35)24-13-9-22(10-14-24)27-20-33-37-28(31)26(19-32-29(27)37)21-7-11-23(12-8-21)34-30(38)39-25-5-3-2-4-6-25/h2-14,19-20H,15-18,31H2,1H3,(H,34,38) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 37 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK in human Jurkat cells assessed as ZAP70 phosphorylation by FACS assay |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase HCK
(Homo sapiens (Human)) | BDBM50320225

(CHEMBL1085316 | N-(4-(7-amino-3-(3-(4-methylpipera...)Show SMILES CN1CCN(CC1)c1cccc(c1)-c1cnn2c(N)c(cnc12)-c1ccc(NC(=O)CCC2CCCC2)cc1 Show InChI InChI=1S/C31H37N7O/c1-36-15-17-37(18-16-36)26-8-4-7-24(19-26)28-21-34-38-30(32)27(20-33-31(28)38)23-10-12-25(13-11-23)35-29(39)14-9-22-5-2-3-6-22/h4,7-8,10-13,19-22H,2-3,5-6,9,14-18,32H2,1H3,(H,35,39) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 37 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of HCK |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320219
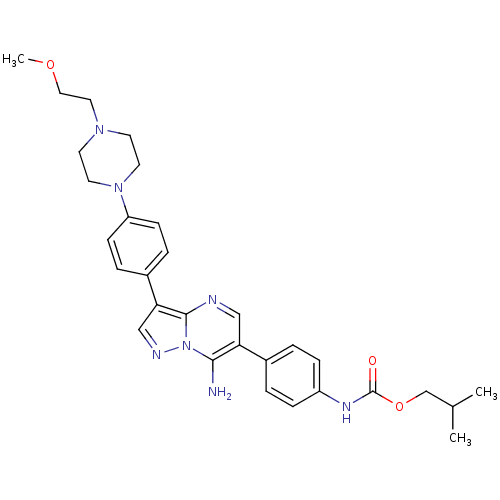
(CHEMBL1082958 | isobutyl4-(7-amino-3-(4-(4-(2-meth...)Show SMILES COCCN1CCN(CC1)c1ccc(cc1)-c1cnn2c(N)c(cnc12)-c1ccc(NC(=O)OCC(C)C)cc1 Show InChI InChI=1S/C30H37N7O3/c1-21(2)20-40-30(38)34-24-8-4-22(5-9-24)26-18-32-29-27(19-33-37(29)28(26)31)23-6-10-25(11-7-23)36-14-12-35(13-15-36)16-17-39-3/h4-11,18-19,21H,12-17,20,31H2,1-3H3,(H,34,38) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 37 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK in human Jurkat cells assessed as ZAP70 phosphorylation by FACS assay |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase ABL1
(Homo sapiens (Human)) | BDBM50320203

(CHEMBL1085511 | isobutyl4-(7-amino-3-(4-(4-methylp...)Show SMILES CC(C)COC(=O)Nc1ccc(cc1)-c1cnc2c(cnn2c1N)-c1ccc(cc1)N1CCN(C)CC1 Show InChI InChI=1S/C28H33N7O2/c1-19(2)18-37-28(36)32-22-8-4-20(5-9-22)24-16-30-27-25(17-31-35(27)26(24)29)21-6-10-23(11-7-21)34-14-12-33(3)13-15-34/h4-11,16-17,19H,12-15,18,29H2,1-3H3,(H,32,36) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 40 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of ABL |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Cathepsin B
(Homo sapiens (Human)) | BDBM50135536
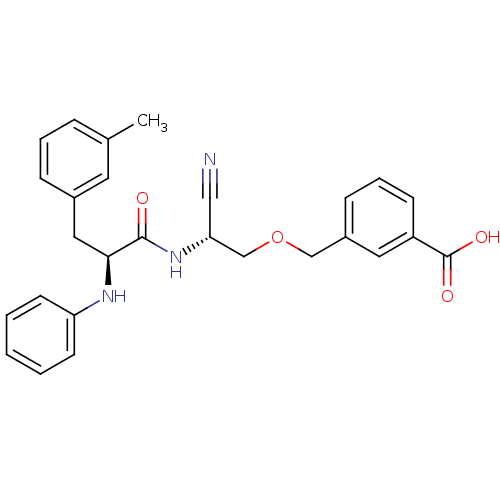
(3-[(R)-2-Cyano-2-((S)-2-phenylamino-3-m-tolyl-prop...)Show SMILES Cc1cccc(C[C@H](Nc2ccccc2)C(=O)N[C@@H](COCc2cccc(c2)C(O)=O)C#N)c1 Show InChI InChI=1S/C27H27N3O4/c1-19-7-5-8-20(13-19)15-25(29-23-11-3-2-4-12-23)26(31)30-24(16-28)18-34-17-21-9-6-10-22(14-21)27(32)33/h2-14,24-25,29H,15,17-18H2,1H3,(H,30,31)(H,32,33)/t24-,25+/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 49 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibtitory activity against cathepsin B (catB) |
Bioorg Med Chem Lett 13: 4121-4 (2003)
BindingDB Entry DOI: 10.7270/Q2BC3XZC |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320221

(CHEMBL1085317 | isobutyl4-(7-amino-3-(3-(4-methylp...)Show SMILES CC(C)COC(=O)Nc1ccc(cc1)-c1cnc2c(cnn2c1N)-c1cccc(c1)N1CCN(C)CC1 Show InChI InChI=1S/C28H33N7O2/c1-19(2)18-37-28(36)32-22-9-7-20(8-10-22)24-16-30-27-25(17-31-35(27)26(24)29)21-5-4-6-23(15-21)34-13-11-33(3)12-14-34/h4-10,15-17,19H,11-14,18,29H2,1-3H3,(H,32,36) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 63 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK in human Jurkat cells assessed as ZAP70 phosphorylation by FACS assay |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase HCK
(Homo sapiens (Human)) | BDBM50320224

(1-(4-(7-amino-3-(3-(4-methylpiperazin-1-yl)phenyl)...)Show SMILES CN1CCN(CC1)c1cccc(c1)-c1cnn2c(N)c(cnc12)-c1ccc(NC(=O)NCC2CCCCC2)cc1 Show InChI InChI=1S/C31H38N8O/c1-37-14-16-38(17-15-37)26-9-5-8-24(18-26)28-21-35-39-29(32)27(20-33-30(28)39)23-10-12-25(13-11-23)36-31(40)34-19-22-6-3-2-4-7-22/h5,8-13,18,20-22H,2-4,6-7,14-17,19,32H2,1H3,(H2,34,36,40) | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 70 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of HCK |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Platelet-derived growth factor receptor alpha
(Homo sapiens (Human)) | BDBM50320203

(CHEMBL1085511 | isobutyl4-(7-amino-3-(4-(4-methylp...)Show SMILES CC(C)COC(=O)Nc1ccc(cc1)-c1cnc2c(cnn2c1N)-c1ccc(cc1)N1CCN(C)CC1 Show InChI InChI=1S/C28H33N7O2/c1-19(2)18-37-28(36)32-22-8-4-20(5-9-22)24-16-30-27-25(17-31-35(27)26(24)29)21-6-10-23(11-7-21)34-14-12-33(3)13-15-34/h4-11,16-17,19H,12-15,18,29H2,1-3H3,(H,32,36) | PDB
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 74 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of PDGFRalpha |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320206
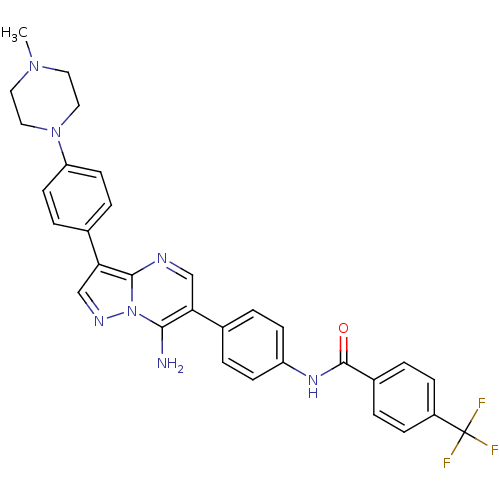
(CHEMBL1085733 | N-(4-(7-amino-3-(4-(4-methylpipera...)Show SMILES CN1CCN(CC1)c1ccc(cc1)-c1cnn2c(N)c(cnc12)-c1ccc(NC(=O)c2ccc(cc2)C(F)(F)F)cc1 Show InChI InChI=1S/C31H28F3N7O/c1-39-14-16-40(17-15-39)25-12-6-21(7-13-25)27-19-37-41-28(35)26(18-36-29(27)41)20-4-10-24(11-5-20)38-30(42)22-2-8-23(9-3-22)31(32,33)34/h2-13,18-19H,14-17,35H2,1H3,(H,38,42) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 75 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |
Tyrosine-protein kinase Lck
(Homo sapiens (Human)) | BDBM50320205

(CHEMBL1083001 | N-(4-(7-amino-3-(4-(4-methylpipera...)Show SMILES CN1CCN(CC1)c1ccc(cc1)-c1cnn2c(N)c(cnc12)-c1ccc(NC(=O)CCC2CCCC2)cc1 Show InChI InChI=1S/C31H37N7O/c1-36-16-18-37(19-17-36)26-13-9-24(10-14-26)28-21-34-38-30(32)27(20-33-31(28)38)23-7-11-25(12-8-23)35-29(39)15-6-22-4-2-3-5-22/h7-14,20-22H,2-6,15-19,32H2,1H3,(H,35,39) | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 76 | n/a | n/a | n/a | n/a | n/a | n/a |
Novartis Institute of Biomedical Research
Curated by ChEMBL
| Assay Description
Inhibition of LCK in human Jurkat cells assessed as ZAP70 phosphorylation by FACS assay |
Bioorg Med Chem Lett 20: 3628-31 (2010)
Article DOI: 10.1016/j.bmcl.2010.04.112
BindingDB Entry DOI: 10.7270/Q2XP754D |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data